Field actions happen; even to the most diligent organizations.
Organizations across the medical device manufacturing industry understand this as a sharp reality. Over the last decade, however, product recalls have substantially increased and are reaching record highs. Over 208 million devices were recalled in the beginning of 2018, more than the total number of recalled devices in all of 2017.

The medical device recall trend is expected to continue to increase in response to the FDA accepting devices with artificial intelligence, higher regulatory requirements, supply chain complexities, increasing component sophistication, and improved testing technologies. Approximately 55 percent of medical device recalls were nationwide in Q1 of 2018.2
Despite the expanding occurrence of product recalls, there have been limited new technologies or innovative management techniques developed in this space. Still, many of the challenges remain universal.
When faced with a product recall, manufacturers often find themselves asking the following questions:
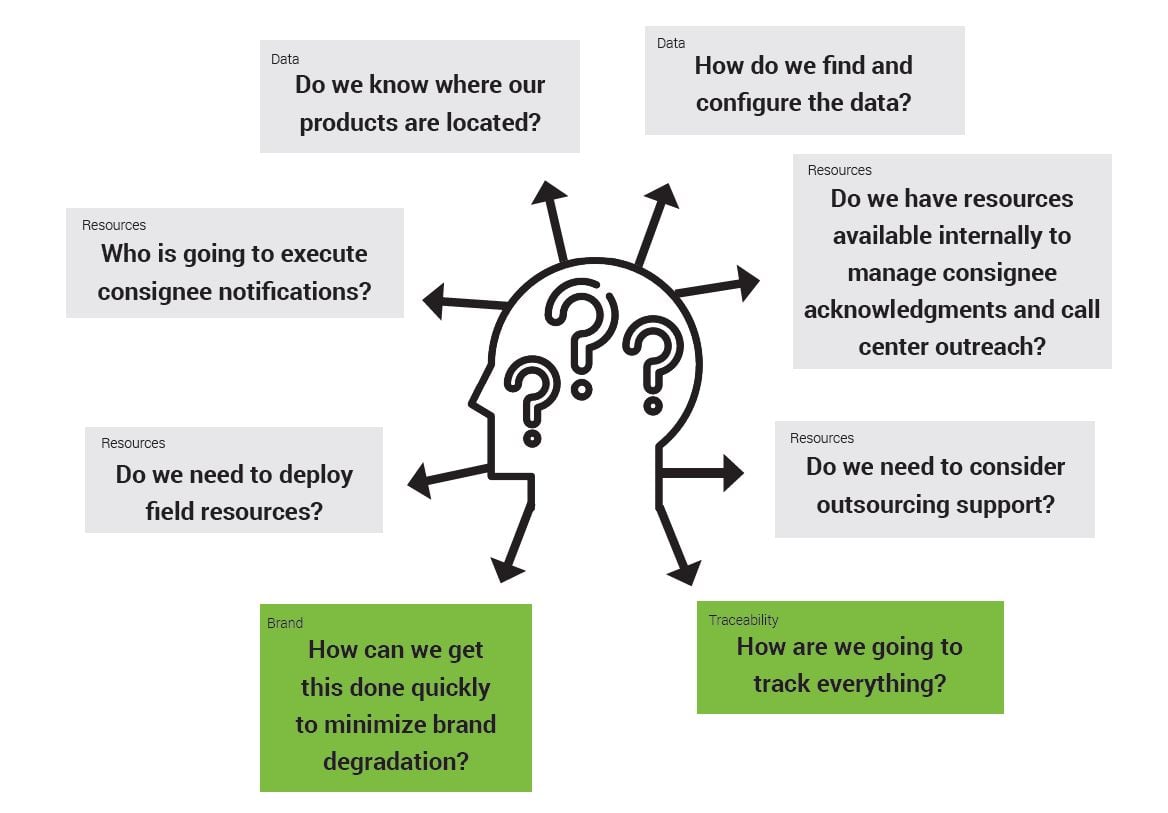
To answer these questions, manufacturers often rely on internal intel or apply previous recall strategies to the new field action.
If you don't know how to answer these questions or are interested in learning more about how new technology is simplifying the recall process for manufacturers, click on the link below or watch this 60-second video.